Managing Supplier Quality Audits – PowerPoint PPTX Template
PowerPoint (PPTX) 64 Slides
BENEFITS OF THIS DOWNLOADABLE POWERPOINT DOCUMENT
- Provides a framework for supplier quality auditing.
- Provides guidance on how to conduct a supplier quality audit professionally and systematically.
AUDIT MANAGEMENT PPT TEMPLATE DESCRIPTION
The purpose of a Supplier Quality Audit is for you to verify if the supplier's quality systems, processes, or products satisfy or are in compliance with the requirements as set forth in the contractual agreements, procedures, and/or agreed upon standards.
This presentation is specially designed for professionals related to Supply Chain, Purchasing, Quality Assurance and Manufacturing Management who are responsible for managing your supplier's quality performance as well as those who are interested in developing and enhancing their personal competence and effectiveness in Supplier Quality Auditing.
LEARNING OBJECTIVES
1. Acquire knowledge on the principles of management system auditing
2. Define the objectives of supplier quality audit
3. Describe how to plan and prepare for a supplier audit
4. Explain the process of on-site supplier audit
5. Describe the post-audit activity
CONTENTS
1. Principles of Management System Auditing
2. Introduction and Objectives of Supplier Quality Audit
3. Supplier Audit Planning and Preparation
4. On-site Supplier Audit
5. Post-audit Activities
6. Do's and Don'ts
Appendix – Additional Information for Supplier Audit
Got a question about the product? Email us at support@flevy.com or ask the author directly by using the "Ask the Author a Question" form. If you cannot view the preview above this document description, go here to view the large preview instead.
PRESENTATION DEEP DIVE ANALYSIS
This deep-dive analysis was generated from the full 64-slide PowerPoint presentation.
Executive Summary
The "Managing Supplier Quality Audits" presentation is an essential resource for organizations aiming to enhance their supplier quality management processes. Developed by Operational Excellence Consulting, this training deck provides a structured approach to conducting effective supplier audits. It covers the principles of management system auditing, planning and preparation for audits, on-site audit processes, and post-audit activities. By utilizing this presentation, organizations can ensure their suppliers consistently deliver high-quality products and services, thereby minimizing risks and enhancing overall operational efficiency.
Who This Is For and When to Use
• Quality Assurance Managers focused on supplier performance
• Procurement Teams responsible for supplier selection and evaluation
• Operations Managers overseeing supply chain reliability
• Compliance Officers ensuring regulatory adherence in supplier processes
Best-fit moments to use this deck:
• When onboarding new suppliers to assess their quality systems
• During performance evaluations of existing suppliers to identify improvement areas
• In preparation for critical supplier audits to ensure compliance and quality standards
Learning Objectives
• Acquire knowledge on the principles of management system auditing
• Define the objectives of supplier quality audits
• Describe how to plan and prepare for a supplier audit
• Explain the process of conducting on-site supplier audits
• Describe the activities following an audit to ensure compliance and improvement
Table of Contents
• Principles of Management System Auditing (page 3)
• Introduction and Objectives of Supplier Quality Audit (page 5)
• Supplier Audit Planning and Preparation (page 26)
• On-site Supplier Audit (page 33)
• Post-audit Activities (page 39)
• Do’s and Don’ts (page 48)
• Appendix - Additional Information for Supplier Audit (page 57)
Primary Topics Covered
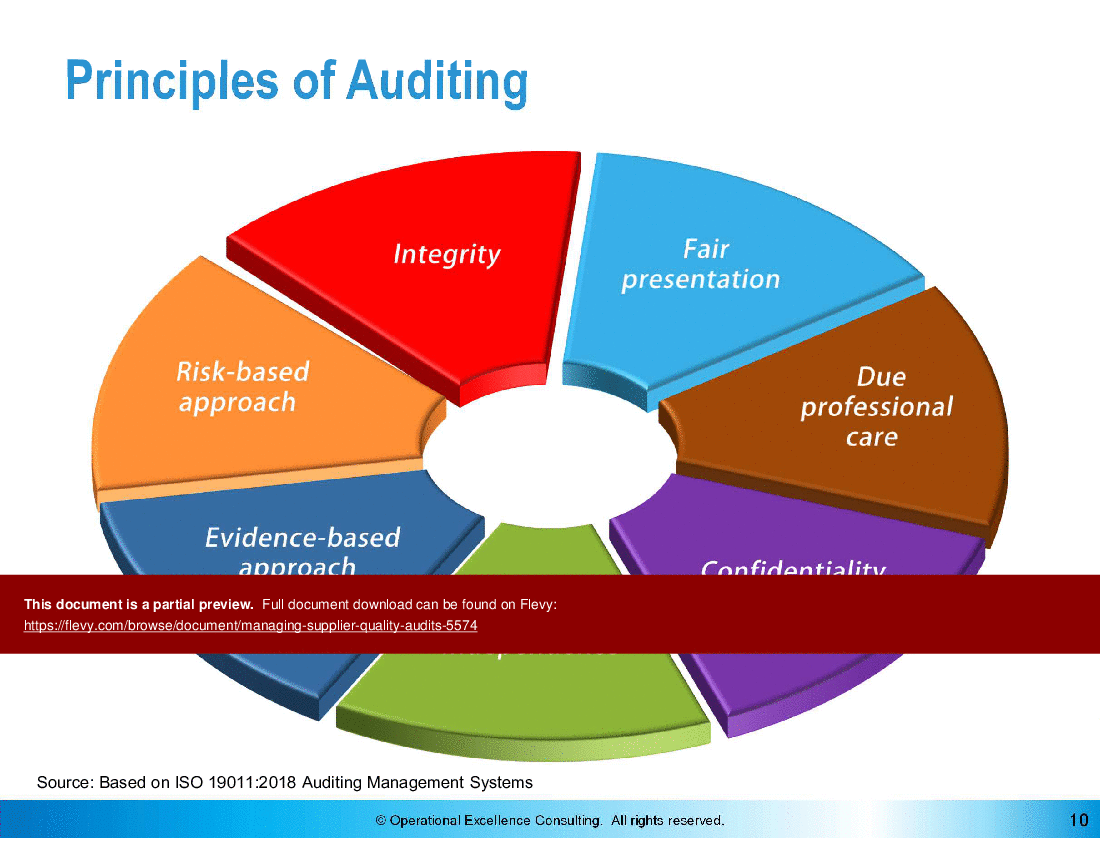
• Principles of Auditing - Understanding the foundational principles that guide effective auditing practices, including integrity, independence, and evidence-based approaches.
• Supplier Quality Audit Objectives - Identifying the key goals of conducting supplier audits, such as assessing management practices and quality systems.
• Audit Planning and Preparation - Steps to effectively plan and prepare for supplier audits, including team selection and pre-audit meetings.
• On-site Audit Process - Detailed procedures for conducting supplier audits, including interviews and documentation reviews.
• Post-audit Activities - Actions to be taken after the audit, including reporting findings and establishing corrective action plans.
• Do’s and Don’ts of Auditing - Best practices and common pitfalls to avoid during supplier audits to ensure effectiveness and professionalism.
Deliverables, Templates, and Tools
• Supplier audit planning checklist template
• On-site audit documentation review guide
• Post-audit report template for findings and corrective actions
• Corrective action plan template for suppliers
• Audit team roles and responsibilities matrix
• Risk assessment framework for supplier evaluation
Slide Highlights
• Overview of the principles of auditing, emphasizing the importance of integrity and independence
• Visual representation of the audit process, detailing each step from planning to post-audit activities
• Key objectives of supplier audits, highlighting their role in ensuring quality and compliance
• Do's and Don'ts slide that succinctly outlines best practices for conducting audits
• Appendix slide summarizing additional resources and methodologies for effective supplier audits
Potential Workshop Agenda
Supplier Audit Planning Session (90 minutes)
• Discuss the objectives and scope of the upcoming supplier audit
• Review the audit team roles and responsibilities
• Establish a timeline and communication plan for the audit
On-site Audit Execution (Half-day)
• Conduct interviews with supplier personnel and review documentation
• Observe processes and assess compliance with quality standards
• Gather evidence and document findings
Post-audit Review and Action Planning (60 minutes)
• Present audit findings to supplier management
• Develop a corrective action plan with timelines for implementation
• Schedule follow-up meetings to assess progress on corrective actions
Customization Guidance
• Tailor the audit planning checklist to align with specific supplier categories and risk levels
• Adjust the post-audit report template to reflect organizational branding and reporting standards
• Incorporate specific regulatory requirements relevant to your industry into the audit process
Secondary Topics Covered
• Types of supplier quality audits, including new supplier assessments and follow-up audits
• Importance of a risk-based approach in audit planning and execution
• Techniques for effective communication during the audit process
• Strategies for engaging suppliers in continuous improvement efforts
Topic FAQ
What are the core principles that guide management system auditing for suppliers?
Management system auditing for suppliers is guided by principles such as integrity, independence, and an evidence-based approach, which ensure auditable findings are verifiable and impartial. These principles are covered in the presentation’s Principles of Management System Auditing section on page 3.What steps should I follow when planning a supplier quality audit?
Planning typically includes defining objectives, selecting the audit team, holding pre-audit meetings, and establishing scope and timelines. The Managing Supplier Quality Audits deck explicitly addresses audit planning and preparation and includes a supplier audit planning checklist template found on page 26.How is an on-site supplier audit usually conducted?
On-site supplier audits involve interviewing personnel, reviewing documentation, observing processes, and collecting evidence to assess compliance and performance. The presentation details these procedures in the On-site Supplier Audit section and provides an on-site documentation review guide on page 33.What post-audit activities should ensure findings are acted upon?
Post-audit activities include reporting findings, developing corrective action plans with timelines, and scheduling follow-ups to verify implementation. Managing Supplier Quality Audits provides a post-audit report template and a corrective action plan template to support these steps, referenced on page 39.How often should supplier audits be scheduled for different supplier types?
Audit frequency depends on supplier performance and criticality; new or critical suppliers typically require more frequent audits, while stable, well-performing suppliers may be audited less often. The overview notes frequency varies by supplier criticality and performance level.What should I look for when purchasing a supplier audit template or training deck?
Look for materials that map to the full audit lifecycle—principles, objectives, planning, on-site execution, and post-audit follow-up—and include practical artifacts such as checklists, report templates, and corrective action templates. Managing Supplier Quality Audits includes these deliverables, including a planning checklist template.I need to onboard a critical supplier quickly—what audit approach is appropriate?
For rapid onboarding of a critical supplier, use a risk-based planning approach to prioritize high-risk processes, perform focused on-site documentation and process observations, and require immediate corrective action plans for any gaps. The presentation recommends a risk-based approach and provides a risk assessment framework.My supplier repeatedly fails specifications—how can audits drive improvement?
Use audits to diagnose root causes through evidence-based review, document non-conformances, require corrective action plans with timelines, and schedule follow-up audits to verify effectiveness. The deck outlines post-audit activities and includes a corrective action plan template and guidance for follow-up audits.Document FAQ
These are questions addressed within this presentation.
What is the purpose of a supplier quality audit?
A supplier quality audit aims to assess a supplier's management practices and quality systems to ensure they meet organizational standards and regulatory requirements.
When should a supplier audit be conducted?
Supplier audits should be conducted when onboarding new suppliers, when existing suppliers show performance issues, or when a supplier is critical to supply chain reliability.
What are the key steps in conducting a supplier audit?
Key steps include planning and preparation, conducting the on-site audit, and performing post-audit activities to ensure corrective actions are taken.
How can I ensure the audit findings are actionable?
Ensure that the audit report clearly outlines findings, corrective actions required, and timelines for implementation, allowing suppliers to respond effectively.
What are the common pitfalls to avoid during supplier audits?
Avoid treating audits as mere paperwork exercises, requiring immediate solutions, or failing to provide constructive recommendations for improvement.
How often should supplier audits be conducted?
Frequency depends on supplier performance and criticality; new suppliers may require more frequent audits, while well-performing suppliers may be audited annually.
What documentation is typically reviewed during a supplier audit?
Auditors review quality manuals, organizational charts, quality control policies, and any relevant process documentation.
How can I prepare my team for conducting supplier audits?
Training on auditing principles, effective communication, and specific supplier processes is essential for audit team readiness.
Glossary
• Supplier Quality Audit - A systematic examination of a supplier's quality management system and practices.
• Corrective Action Plan - A document outlining steps to address identified deficiencies.
• Risk-based Approach - An auditing strategy focusing on areas of highest risk to ensure effective resource allocation.
• On-site Audit - An audit conducted at the supplier's location to assess processes and compliance.
• Audit Team - A group of individuals responsible for conducting the audit, typically including a lead assessor and support members.
• Non-conformance - A failure to meet specified requirements during the audit process.
• Evidence-based Approach - A method of auditing that relies on verifiable evidence to support findings.
• Supplier Management Review - A process for evaluating supplier performance and addressing issues identified during audits.
• Quality Management System - A structured system that documents processes, procedures, and responsibilities for achieving quality policies and objectives.
• Follow-up Audit - An audit conducted to verify the effectiveness of corrective actions taken by the supplier.
• ISO Standards - International standards that outline best practices for quality management systems.
• Documentation Review - The process of examining supplier documents to assess compliance with quality standards.
• Supplier Assessment - An evaluation of a supplier's capabilities and performance metrics.
• Continuous Improvement - Ongoing efforts to enhance products, services, or processes.
• Quality Control Manual - A document that outlines the quality control processes and procedures within an organization.
• Supplier Performance Metrics - Quantitative measures used to assess a supplier's quality and delivery performance.
• Audit Findings - The results of the audit process, including identified strengths and weaknesses.
• Supplier Engagement - The process of involving suppliers in quality improvement initiatives.
• Quality Methodologies - Various approaches to quality management, such as Lean and Six Sigma.
• Sampling Arrangements - The strategy used to select samples for quality inspection during audits.
• Inspection Process - The procedures used to evaluate products or services for compliance with quality standards.
Source: Best Practices in Audit Management, Supplier Management PowerPoint Slides: Managing Supplier Quality Audits PowerPoint (PPTX) Presentation Slide Deck, Operational Excellence Consulting